Research Results of New Biomarker for Ovarian Cancer Diagnosis through Bertis’ Platform Published in Journal of Proteome Research
Presentation of a new multi-biomarker panel with 95% accuracy for stage 1 to 3 ovarian cancer based on targeted mass spectrometry (MS) and bioinformatics
Research results through Bertis’ technology platform were published as a cover article in the September issue of the international scientific journal, Journal of Proteome Research
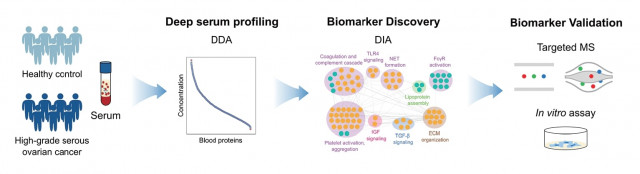
Discovery process of high-grade serous ovarian cancer (HGSOC) biomarker candidates
SEONGNAM--(뉴스와이어)--Bertis, a proteomics-based precision medicine technology development company (CEOs Dong-young Noh, Seung-man Han), announced that the research results of their search for protein biomarkers for early diagnosis of high-grade serous ovarian cancer (HGSOC) were published in the September issue of the international scientific journal, Journal of Proteome Research.[1]
Ovarian cancer is the first leading cause of death among gynecological cancers. Most patients are asymptomatic in early stages, and as effective early diagnosis methods have not yet been secured, 70% of the cases are not detected until the cancer develops into stage 3 or higher.[2, 3] After stage 3, the survival rate of the patient is drastically reduced. The 5-year survival rate is more than 76% in stage 1 and approximately 60% to 74% in stage 2, while the 5-year survival rate in early stage 3 is known to be 41%, 23% in late stage 3, and just 11% in stage 4.[4] In particular, HGSOC is the most common type of ovarian cancer, accounting for 70% of all ovarian cancers. For decades, the overall survival rate of HGSOC has barely improved, and the need for the introduction of a standard testing method for early diagnosis has been emphasized.[5, 6]
The research team at Bertis presented a new multi-biomarker panel candidate with 95% accuracy (100% sensitivity, 91% specificity) for the diagnosis of stage 1 to 3 HGSOC. The results of this research were published in the September issue of the Journal of Proteome Research on the 2nd by the American Chemical Society. In particular, the results were included as one of the supplementary cover articles in this issue with 16 papers listed.
In this study, the research team identified 18 candidate proteins after quantifying 1,847 serum proteins, the highest number ever reported as ovarian cancer biomarker research results, through Bertis’ technology platform to discover biomarker candidates. As a result of developing and evaluating a predictive model, the accuracy of the diagnostic value for stages 1 to 3[7] HGSOC was 95% (sensitivity 100%, specificity 91%) when 18 candidate proteins were combined with multiple biomarkers.
Un-beom Kang, Head of Bertis Biomarker Research Institute, who led this study, explained, “This study is significant in that it secured the results of in-depth blood proteome research based on Bertis’ advanced proteomics analysis technology, as well as presented a new biomarker panel with high accuracy for ovarian cancer,” and added that, “We expect that based on data from this study, research and development to provide standardized testing for early diagnosis of ovarian cancer will be expedited and the creation of results will be accelerated.”
“Proteomics analysis is an incredibly effective tool for understanding cancer and discovering new biomarkers for diagnosis and prognosis,” said, Seung-man Han, Bertis’ CEO. “As a company that has led the discovery of biomarkers based on proteomics technology as well as the development of clinical solutions using it, we will do our best to contribute to the improvement of the diagnosis and treatment of major diseases through continuous research and development.”
Bertis is a company that combines proteomics and bioinformatics to develop biomarkers for cancer and other various major diseases, as well as provides diagnosis and analysis solutions. It succeeded in commercializing proteomics technology with Mastocheck, the world’s first proteomics-based blood testing solution for early breast cancer diagnosis, and PASS (Pan-omics Analysis Service & Solution), an integrated analysis solution for Panomics. Currently, Bertis is researching and developing early diagnosis solutions for pancreatic cancer, ovarian cancer, etc.
About Bertis
Bertis is a company that has discovered biomarkers for cancer and other major disorders and provided diagnostic and analysis solutions based on the combination of proteomics and bioinformatics. Since our establishment in 2014, our mission has been to contribute to the health and happy life of humanity through the realization of early diagnosis medicine with a focus on the most advanced quantitative proteomics research, precision medicine based on companion diagnostic markers, and companion diagnostics for oncology drug development and personalized treatment. Our flagship solutions include Mastocheck, the world’s first proteomics-based blood test solution for early breast cancer detection, and PASS (Pan-omics Analysis Service & Solution), a platform for integrative analysis of pan-omics (multi-omics) data. For more information, please visit our website www.bertis.com or follow us on LinkedIn (www.linkedin.com/company/bertis-inc).
[1] Sunghyun Huh, et al. Novel Diagnostic Biomarkers for High-Grade Serous Ovarian Cancer Uncovered by Data-Independent Acquisition Mass Spectrometry. Journal of Proteome Research. Available at https://pubs.acs.org/doi/10.1021/acs.jproteome.2c00218 (accessed on August 9, 2022)
[2] Lim MC et al. Survival After Hyperthermic Intraperitoneal Chemotherapy and Primary or Interval Cytoreductive Surgery in Ovarian Cancer. JAMA Surg. 2022;157(5):374-383. doi:10.1001/jamasurg.2022.0143 Available at https://jamanetwork.com/journals/jamasurgery/fullarticle/2789724 (accessed on August 12, 2022)
[3] Medical information about ovarian cancer on Korean Society of Obstetrics and Gynecology official website. Available at https://www.ksog.org/public/index.php?sub=4&third=6
[4] Cancer Information Service; ovarian cancer on National Cancer Information Center official website. Available at https://www.cancer.go.kr/lay1/program/S1T211C223/cancer/view.do?cancer_seq=3581&menu_seq=3600 (accessed on August 12, 2022)
[5] Hanash, S. M.; Pitteri, S. J.; Faca, V. M. Mining the Plasma Proteome for Cancer Biomarkers. Nature 2008, 452, 571−579.
[6] Zheng, H.; Zhang, L.; Zhao, Y.; Yang, D.; Song, F.; Wen, Y.; Hao, Q.; Hu, Z.; Zhang, W.; Chen, K. Plasma MiRNAs as Diagnostic and Prognostic Biomarkers for Ovarian Cancer. PLoS One 2013, 8, No. e77853.
[7] Federation of Gynecology and Obstetrics (FIGO) standard stage